First COVID-19 vaccine could be delivered to distribution points as early as end of December, Fortin says

Federal officials today explained how they plan to roll out millions of COVID-19 vaccine doses in the coming weeks as Ottawa launches its mass inoculation campaign.
Janssen seeks Health Canada approval for its COVID-19 vaccine
The initial supply of the doses will be limited — just three million Canadians are expected to get shots in the first three months of 2021. Millions more doses are expected to arrive as the supply chain stabilizes.
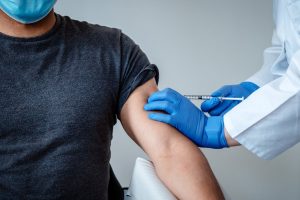
One of the principal challenges facing the immunization effort is the distribution of vaccines that must be kept at very low temperatures – well below those that a standard commercial refrigerator can offer.
The Pfizer vaccine, which is expected to get the green light from Health Canada as early as this month, needs to be kept at approximately -80 degrees Celsius to remain stable. The Moderna product, another vaccine that uses groundbreaking messenger RNA (mRNA) technology, must be kept at -20 degrees Celsius.
Maj.-Gen. Dany Fortin, a former NATO commander in Iraq, is leading vaccination logistics at a new national operations centre in the Public Health Agency of Canada.
While the country is facing unprecedented “logistical complexities,” he said, the military and its partners will be ready to deploy vaccines as soon as they are approved in Canada.
Fortin said the national operations centre isn’t waiting for Health Canada’s sign-off to begin preparations.
The Pfizer product will be delivered by that company directly to provincial and territorial distribution points as early as the end of the month. The federal government already has secured the cold storage required for this product.
All of the provinces have indicated where the Pfizer-specific fridges should be placed and 14 distribution points nationwide will be ready to receive the vaccine starting on Dec. 14, Fortin said.
A senior official, speaking to CBC News on a not-for-attribution basis, said Alberta, B.C., Ontario and Quebec will get two such delivery sites each, with one in each of the other provinces. A plan for the territories is still being finalized, the official said.
The shots likely will be distributed on a per capita basis, the official said, much like how federally procured personal protective equipment has been issued to those jurisdictions throughout this pandemic. Some observers have said provinces dealing with higher caseloads should get priority access to vaccine shots at first.
Manitoba Premier Brian Pallister said Thursday a per-capita distribution plan will disadvantage his hard-hit province. He also raised concerns about Ottawa’s plan to take the lead on inoculating Indigenous people. “This hurts Manitobans, to put it mildly,” he told reporters.
Eventually, there will be 205 “points of issue” locations across the country where health care professionals can administer the vaccine, Fortin said. It will be up to the provinces and territories to specify where and when individual Canadians will be inoculated.
Fortin said at least one “dry run” has been executed so far, with more planned in the days ahead, to ensure things run smoothly once this vaccine hits our shores from manufacturing hubs in the U.S. and abroad. These practice runs will ensure officials are comfortable with what Fortin called the “very unique requirements” of this Pfizer vaccine.
Preparing for the worst
Fortin said he’s actively planning for multiple worst-case scenarios, such as bad weather, cyber attacks and fires at distribution hubs. IBM, the information technology company, said in a blog post published on Thursday that it had uncovered “a global phishing campaign” targeting the U.S. COVID-19 vaccine distribution mission, Operation Warp Speed.
“We’re very much executing a whole-of-nation approach. The size and scope and scale of this problem is unprecedented and there’s a number of factors at play,” Fortin said.
“I like the idea of being ready before the Christmas timeframe, so we are certain to be ready when it comes in January.”
The general said his team is in daily contact with Pfizer and the company is “comfortable” with the plan that Canada has crafted. Pfizer has said it won’t ship product to a country that isn’t ready to receive a vaccine that is so temperature-sensitive.
Pfizer review ‘progressing really well’: Health Canada
Dr. Supriya Sharma, the chief medical adviser at Health Canada, said Thursday that the regulatory review of Pfizer’s vaccine is “progressing really well” and her department has the “majority of information” it needs from the company to certify that it’s safe and effective.
In an interview with CBC’s Power & Politics, Sharma said the final approval could come in the next 7 to 10 days. The U.S. Food and Drug Administration is set to meet on Dec. 10 to decide on an emergency use authorization (EUA) for that shot and Sharma said Canada is following a similar timeline.
Canada has placed orders with Pfizer and its German partner BioNTech for 20 million doses of the two-dose vaccine, with options for millions more in the months to follow. The company has reported its vaccine was 95 per cent effective in preventing COVID-19 among clinical trial participants who had no evidence of prior infection.
Dr. Howard Njoo, Canada’s deputy chief public health officer, said Canadians shouldn’t be fixated on the exact date when Pfizer gets the nod from Canadians regulators.
“I think we shouldn’t be so obsessive with the actual delivery of the vaccines themselves, the dates and so on. I think what’s really important is the fact we’re planning, preparing, doing exercises,” he said.
“We’re doing the dry runs, the soft launch so that once the vaccine technically arrives, everyone will be comfortable, we’ll be trained to actually utilize the vaccine.”
The Moderna vaccine, which is expected to secure regulatory approvals after the Pfizer product, will be imported into Canada by the federal government, largely through private shipping companies. Ottawa will in turn divide up the product for the provinces and territories.
The government is now finalizing “end mile” contracts with logistics firms — the companies that will transport the Moderna vaccines to centres where Canadians can go for a shot.
On Monday, the Massachusetts-based company applied to the FDA for its EUA for the American marketplace. Data from the company’s final clinical trial are encouraging, demonstrating the vaccine is 94.1 per cent effective at preventing COVID-19 and 100 per cent effective at preventing severe cases of the disease.
Two other companies, AstraZeneca and Johnson & Johnson’s pharmaceutical division, Janssen, have also submitted their vaccines for regulatory approval. Sharma said those companies still need to submit “large chunks of information” before a final decision can be made.
Njoo said the federal government is now refining who is best suited to get an early dose of a vaccine — early guidance from the National Advisory Committee on Immunization (NACI) suggests seniors in long-term care homes and frontline health care workers will be among the first to get a shot.
Conservative Leader Erin O’Toole and his party’s health critic Michelle Rempel Garner held a news conference this morning to discuss an opposition day motion that will call on the government to release its plan by Dec. 16.
O’Toole accused the government of failing to provide Canadians with a plan and a timeframe for vaccine distribution.
“Without a concrete timeline for vaccines, businesses won’t have the confidence to reinvest in their operations and rehire Canadians who have been laid off during the pandemic,” he said.
“Without a reliable timeline, or details, provinces have the impossible task of establishing complex supply chains with no lead time.”
The motion calls for a status update on:
- How each type of vaccine will be safely delivered, stored and distributed to Canadians.
- The date on which each vaccine type will be first deployed in Canada and the rate of vaccinations anticipated by month.
- Any planned federal guidance with respect to the deployment of the vaccine by priority group, such as front-line health workers and seniors.
- The plan to distribute the vaccine to Indigenous communities, members of the Canadian Armed Forces and veterans.







Redes Sociais - Comentários